A charge transfer at the origin of the voltage hysteresis
The transition of our societies to a carbon-free world is underway. Among the issues raised by this observation, the question of energy and its storage occupies an important place. Batteries, especially Li-ion, are today seen as the most mature technology to meet expected challenges. Despite their good performance, researchers and manufacturers still want to be able to increase the energy density of materials to meet the expectations of users, in particular in the field of electric mobility.
For several years, researchers have been interested in the potential of Li-rich electrodes whose typical structure is Li1+xTM1-XO2. Indeed, these materials, due to an original mechanism called anionic redox, have a higher theoretical energy density than conventional materials. Unfortunately, in practice, sluggish kinetics, voltage hysteresis and voltage fade compromise the sales of batteries incorporating these materials.
While the battery community accepts the idea that the voltage fade is due to the irreversible migration of cations in Li-rich materials, debates are much sharper as to the origin of the hysteresis. The explanation by the irreversible cationic migration is also proposed in this case but it turns out to be insufficient. For example, Na2RuO3 is a counterexample: The material is the site of a phenomenon of hysteresis without, however, exhibiting cationic migration.
In an article published in Nature Chemistry, an international team coordinated by Prof. Jean-Marie Tarascon tries to explain the origin of this phenomenon and suggests some ways to mitigate it.
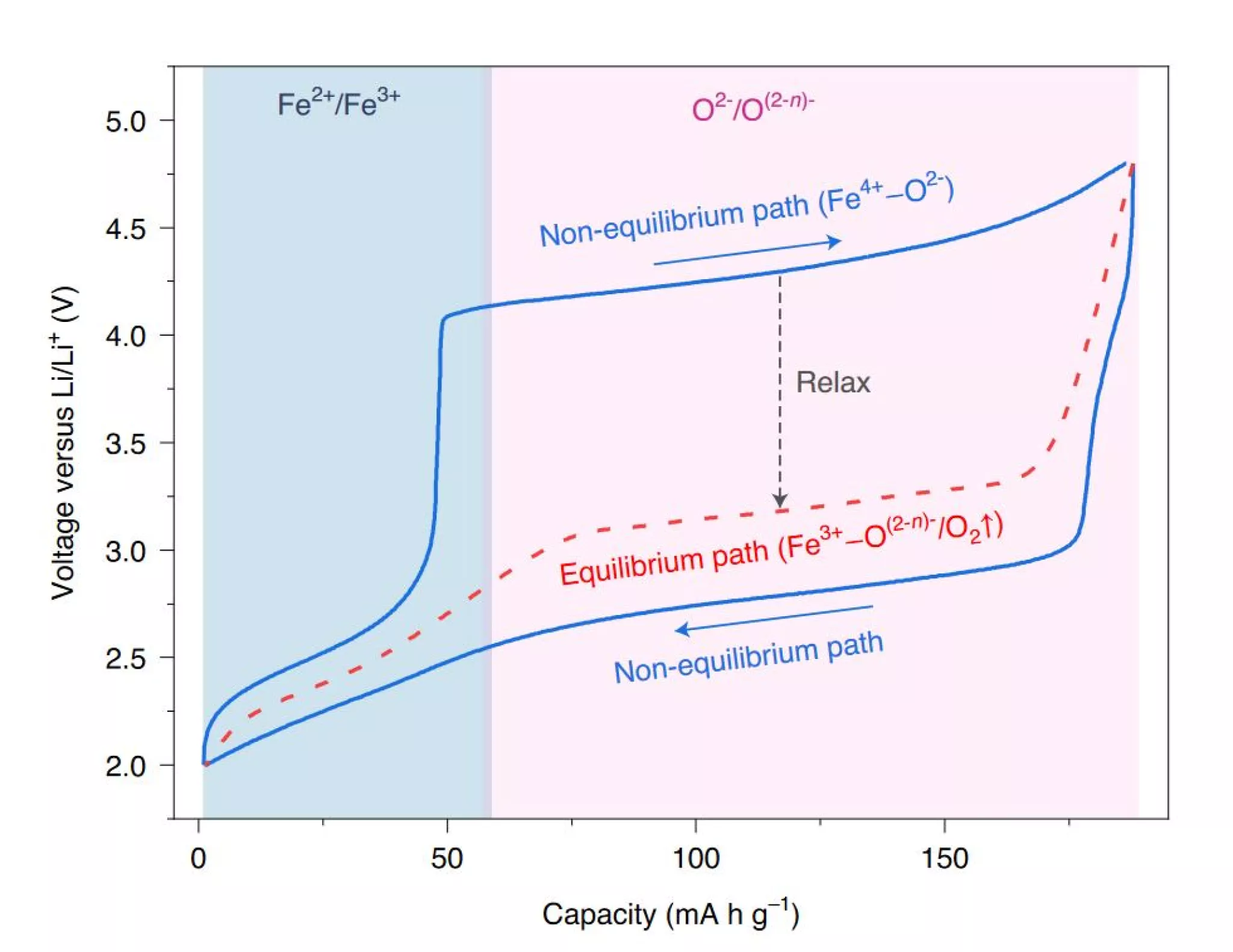
The researchers show that the hysteresis is in fact due to a kinetically slow charge transfer between oxygen atoms (O) and metal atoms (M). This explanation has the merit of being able to be applied more universally to Li-rich materials than the hypotheses previously put forward while avoiding too many exceptions. In addition, the study discussed here shows that hysteresis increases with the degree of polarization of M-O bonds. In short, the intensity of the phenomenon is more pronounced with ionic systems.
Based on these conclusions, the RS2E team suggests two solutions to mitigate the hysteresis. The first is to increase the covalency of the M-O bonds or to use less electronegative anions, i.e. S or Se instead of O. The second option would be to play on the local structure of the electrode to directly transfer electrons from oxygen without involving cationic intermediates, which researchers are currently doing.
The study and its conclusions can be found in detail here.